We have previously written about re-packaged Avastin injections becoming contaminated with silicone oil. Injections of Lucentis may also carry this same risk of silicone contamination.
- Lucentis (generic name: ranibizumab) is manufactured by Genentech, Inc. (which also manufactures Avastin).
Lucentis are prescription medications used to treat various forms of macular degeneration. They can also treat diabetic macular edema, diabetic retinopathy, and myopic choroidal neovascularization. Like Avastin, and Lucentis are anti-VEGF (anti-vascular endothelial growth factor) medications. VEGF is a protein that is one of the causes of leaky blood vessels in the eye. Elyea and Lucentis are thought to work by blocking this protein, hence the name “anti-VEGF” medications.
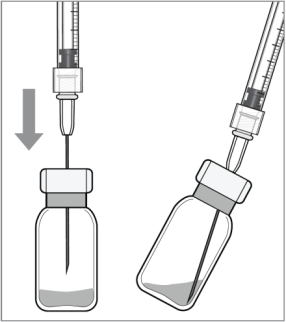
Elyea and Lucentis are administered by an injection into the eye. To be injected into the eye, Elyea and Lucentis must first be placed into a syringe. Both Elyea and Lucentis are delivered to the doctor in small vials and the solution must be drawn out using a syringe (see picture). The needle is then replaced with a smaller needle for injection into the eye.
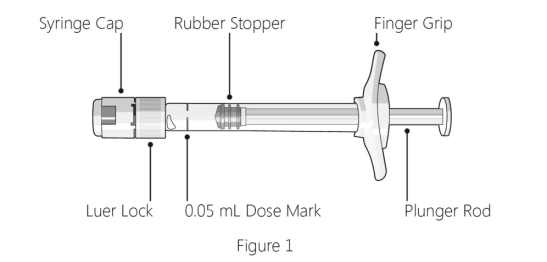
Lucentis is also now available in a pre-filled syringe (seen in picture).
Silicone is used to lubricate the barrel of many syringes. As we have previously reported (here), silicone from the syringe can leach into the solution, which results in silicone droplets / particles being injected into the patient’s eye. If this occurs, the silicone particles can obstruct a person’s vision and severely interfere with daily life.
If you have experienced silicone oil or silicone particles being injected into your eye during an injection of Lucentis, or Avastin, please do not hesitate to contact one of our experienced attorneys at Spangenberg Shibley & Liber. We are actively investigating cases involving silicone contaminated eye injections from various product manufacturers, and we would be happy to review your case personally and discuss with you whether you have a valid claim for compensation. Consultations are free and confidential. Call (216) 600-0114 to request yours.