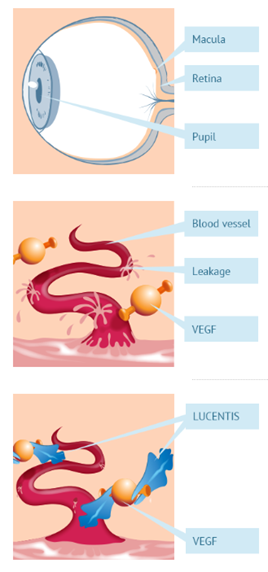
Lucentis and Avastin are all used to treat various forms of macular degeneration. An overproduction of a protein called vascular endothelial growth factor is one of the causes of “leaky” blood vessels, which can lead to vision loss Lucentis, and Avastin are “anti-VEGF” medications, which means they work by blocking this VEGF protein. (See picture from http://www.lucentis.com/information).
Unfortunately, these medications can become contaminated with silicone oil droplets or particles prior to being injected into the eye. The silicone comes from the syringe that is used to inject the solution into the eyes.
Many of the syringes used for injecting Lucentis and Avastin into the eye are manufactured by the giant medical device corporation, Becton Dickinson, and were never approved for use in eye injections.
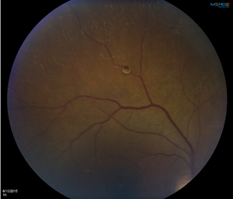
This is a picture of a silicone oil droplet in a patient’s eye caused by a contaminated Avastin injection. This case report was published by Kinker B, et al., on March 27, 2017:
Brenton K J, Asheesh T M, Joaquin T. Intravitreal Silicone Oil Droplets after Multiple Avastin Injections. JOJ Ophthal. 2017; 2(3): 555588. DOI: 10.19080/JOJO.2017.02.555588 (https://juniperpublishers.com/jojo/pdf/JOJO.MS.ID.555588.pdf )
Once a silicone oil droplet or particle is injected into the eye, it can cause serious interference with a person’s vision and daily living.
If you have experienced silicone oil or silicone particulates being injected into your eye during an injection of Lucentis or Avastin, please do not hesitate to contact one of our experienced attorneys at Spangenberg Shibley & Liber LLP. We are actively investigating cases involving silicone contaminated eye injections and we would be happy to review your case personally and discuss with you whether you have a valid claim for compensation. For a free consultation with a member of our legal team, contact us today.